Who discovered Ammonia?
Ammonia was discovered by a man named Fritz Haber, who was a German physical chemist. In 1908, by circulating nitrogen and hydrogen at a pressure of 150-200 atm and at a temperature of about 500°C over the catalyst, he accomplished the synthesis of ammonia after searched for suitable catalysts. This method was developed on a large industrial scale by Carl Bosch, a German industrial chemist. Bosch replaced Haber's catalyst, osmium and uranium into pure iron which would be cheaper and more easily available. This process was discovered in time for the start of World War 1.
Fritz Haber
Fritz Haber was born on December 9, 1868 in Breslau, Germany. He was a German physical chemist, who won the 1918 Nobel Prize of Chemistry for his invention on nitrogen fixation.
As a child, he went to a classic school in Breslau and did a lot of chemical experiments during that time. In 1886, he studied chemistry at the University of Heidelberg, University of Berlin and Technical School at Charlottenberg for 5 years and was educated by different chemists during that time.
Fritz Haber died on January 29, 1934 at Basle because of a grave illness.
Carl Bosch
Carl Bosch was born in August 27, 1874 in Cologne, Germany. He was a German industrial chemist who was known for his development of the Haber-Bosch process for high-pressure synthesis of ammonia. In 1931, Bosch shared the Nobel Prize for chemistry with Friedrich Bergius for their contributions to the invention and development of chemical high- pressure method. He was educated at the Technische Hochschule in Charlottenburg. In 1896, he started reading chemistry at Leipzig University. In 1898
Carl Bosch died on April 26 after an illness when he was 65.
How is it made?
The Haber-Bosch Process
Ammonia was discovered by a man named Fritz Haber, who was a German physical chemist. In 1908, by circulating nitrogen and hydrogen at a pressure of 150-200 atm and at a temperature of about 500°C over the catalyst, he accomplished the synthesis of ammonia after searched for suitable catalysts. This method was developed on a large industrial scale by Carl Bosch, a German industrial chemist. Bosch replaced Haber's catalyst, osmium and uranium into pure iron which would be cheaper and more easily available. This process was discovered in time for the start of World War 1.
Fritz Haber
Fritz Haber was born on December 9, 1868 in Breslau, Germany. He was a German physical chemist, who won the 1918 Nobel Prize of Chemistry for his invention on nitrogen fixation.
As a child, he went to a classic school in Breslau and did a lot of chemical experiments during that time. In 1886, he studied chemistry at the University of Heidelberg, University of Berlin and Technical School at Charlottenberg for 5 years and was educated by different chemists during that time.
Fritz Haber died on January 29, 1934 at Basle because of a grave illness.
Carl Bosch
Carl Bosch was born in August 27, 1874 in Cologne, Germany. He was a German industrial chemist who was known for his development of the Haber-Bosch process for high-pressure synthesis of ammonia. In 1931, Bosch shared the Nobel Prize for chemistry with Friedrich Bergius for their contributions to the invention and development of chemical high- pressure method. He was educated at the Technische Hochschule in Charlottenburg. In 1896, he started reading chemistry at Leipzig University. In 1898
Carl Bosch died on April 26 after an illness when he was 65.
How is it made?
The Haber-Bosch Process
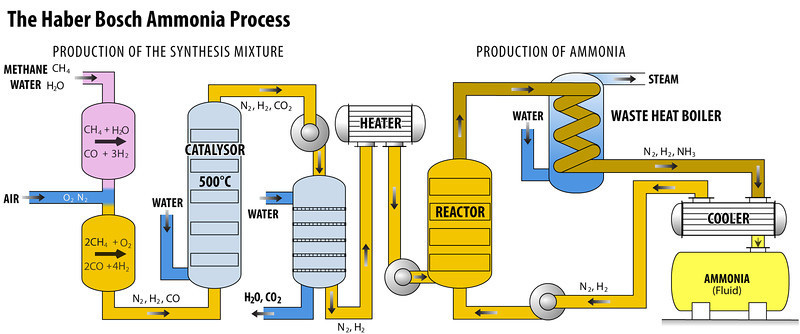
The Haber-Bosch process is a method which synthesizing ammonia by combining nitrogen and hydrogen from natural gas (methane) under high pressure. This was the first industrial chemical process to use high pressure for a chemical reaction. This process requires nitrogen and hydrogen to be fused under high pressure and fairly high temperature. In practical case, they can only be combined under extremely high temperature, thus a catalyst ( mainly made from iron) is used to make this process possible in lower temperature. As soon as it is formed, ammonia is removed form the batch to ensure that an equilibrium favouring product formation is maintained. The lower the temperature and the higher the pressure used, the proportion of ammonia yielded in a mixture.
The Haber-Bosch process was the most important technological advance in the 20th century.